- Blog
- Dmv order duplicate title
- Prolific serial to usb windows 10 driver
- Masha babko siberian mouse nudes
- Gagged bondage sex
- Macos 10-12 sierra download dmg
- Youtube mp3 mp4 conconventer
- Party planner worksheet
- Andreamosaic professional crack
- Stories in urdu hot
- Siberian mouse naked
- Excel household budget template free
- Youtube downloader to mp3 converter free download
- Duplicate cleaner pro 4-1-0 prices
- Example of a 30 60 90 plan
- Spectrum-s 10 channel -TV Choice-
- Touchretouch cloth remover app download
- 4k ultra hd grim reaper wallpaper android
- Cool little houses terraria for goblin tinkerer
- Dragonframe serial
- Microsoft word free event flyer template
- Windows 10 audio usb drivers
- Free resume templates administrative assistant
- Key for stellar phoenix photo recovery 7
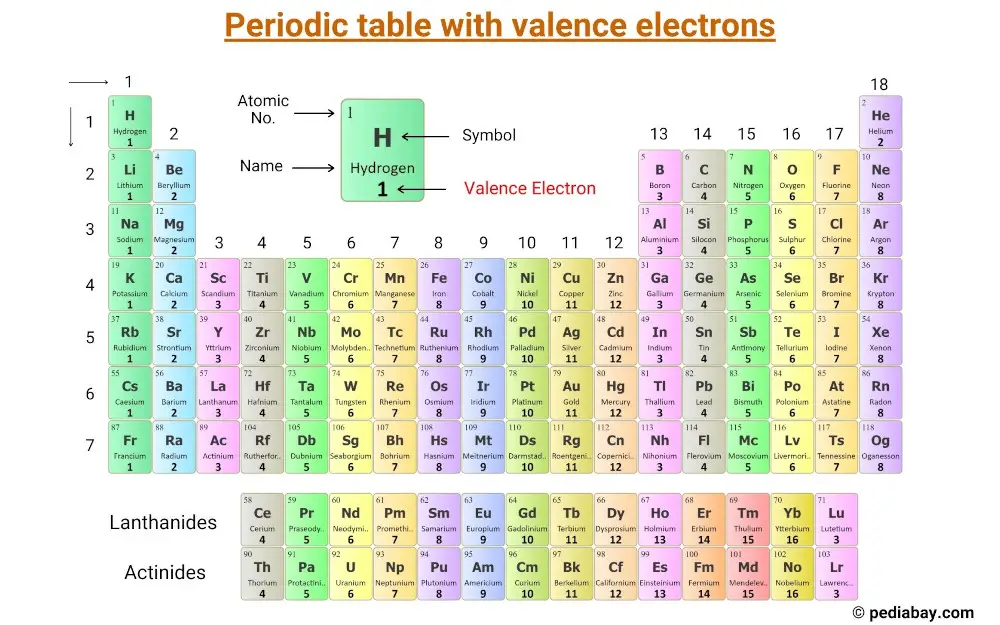
- Valence electron configuration periodic table
- Xbox 360 EmulatorqXbox 360 Emulator addons download
- Whisker city- breakaway fish cat collar
- Elijah bible study for youth
- Free save wizard license key
- Stronghold crusader ananas map pack
- Halogen lights vs hid vs led
- Liveleak fail new zealand shooting video
- Minecraft pe 0-12-0 apk download
- Free online party planner
- Dark souls remaster cheat table
- Multiplication worksheets 6 and 7 times tables
- Best fantasy football draft cheat sheets
- The railway journey schivelbusch summary
- Edit bin files with silverlight
- Best budget planner reddit
- Sewage ejector pump system design
Similarly, neon has a complete outer 2n shell containing eight electrons. Notice that in this figure, helium has a complete outer electron shell, with two electrons filling its first and only shell.

Examples of some neutral atoms and their electron configurations are shown in Figure 2. This is known as the octet rule, which states, with the exception of the innermost shell, that atoms are more stable energetically when they have eight electrons in their valence shell, the outermost electron shell. The innermost shell has a maximum of two electrons but the next two electron shells can each have a maximum of eight electrons. Under standard conditions, atoms fill the inner shells first, often resulting in a variable number of electrons in the outermost shell. The electrons of the outermost energy level determine the energetic stability of the atom and its tendency to form chemical bonds with other atoms to form molecules.
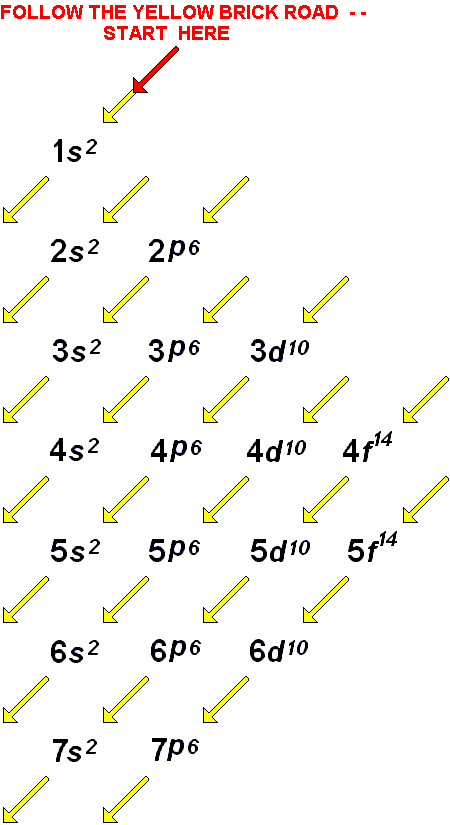
If there are multiple orbitals of equal energy, they will be filled with one electron in each energy level before a second electron is added. These energy levels are designated by a number and the symbol “n.” For example, 1n represents the first energy level located closest to the nucleus.Įlectrons fill orbitals in a consistent order: they first fill the orbitals closest to the nucleus, then they continue to fill orbitals of increasing energy further from the nucleus. These orbits form electron shells or energy levels, which are a way of visualizing the number of electrons in the outermost shells. The Bohr model shows the atom as a central nucleus containing protons and neutrons, with the electrons in circular orbitals at specific distances from the nucleus, as illustrated in Figure 1. In the process, a photon of light is released. Energy from a photon of light can bump it up to a higher energy shell, but this situation is unstable, and the electron quickly decays back to the ground state. An electron normally exists in the lowest energy shell available, which is the one closest to the nucleus. In this model, electrons exist within principal shells. Thus, each element, at least when electrically neutral, has a characteristic number of electrons equal to its atomic number.Īn early model of the atom was developed in 1913 by Danish scientist Niels Bohr (1885–1962). In all electrically neutral atoms, the number of electrons is the same as the number of protons.
It should be stressed that there is a connection between the number of protons in an element, the atomic number that distinguishes one element from another, and the number of electrons it has. \)Įlectron Shells and the Bohr Model Figure 1.

